It is a trial that tests a new treatment against regular or other treatments. Patients of a clinical trial are volunteers and agree to participate only after signing a consent form. Clinical trials are looked after by regulatory agencies (like Swissmedic, FDA) and all data are considered private.
Doctors and scientists of the NISCI trial sites take part in many different kinds of research trials. Some research does not include humans but research that includes humans is called clinical research. Clinical research helps researchers understand how best to treat patients or helps researchers to learn more about a condition or disease. There are many different forms of clinical research. One common form is a clinical trial. In a clinical trial, researchers test new drugs, medical devices or treatments on humans.
After a Spinal Cord Injury (SCI), patients receive rehabilitation to make sure they recover as much as possible. Most patients continue to get better for 3-6 months after injury and sometimes even get a little bit better after that. This is called natural recovery. Before rehabilitation begins and once patients are accepted for the NISCI trial, a computer will decide randomly if a patient gets the new treatment or regular treatment. Neither you nor the trial team will know which treatment you get. This is called a “double blind” trial. A patient who gets regular treatment is called a “control patient”. Control patients are necessary for the success of the trial because we need to compare how much recovery is natural and how much is due to the new treatment. Other than the new treatment, all patients will be cared for exactly the same while you are in-patientand during rehabilitation.
Clinical trials are used to look for new drugs or combination of drugs, new ways of giving the drugs to patients, new types of surgery, new devices, or new treatments. Clinical trials must be done before a new drug, treatment, or device is allowed to be used in hospitals or clinics. The group of people in a country’s government that watch over research is called a regulatory agency. The regulatory agency in the European Union is the European Medicines Agency (EMA). The EMA decides if and how doctors and scientists (Investigators) test a new drug, treatment or device.
Hospitals running clinical trials have groups of people that review all trials at that hospital, called Ethics Committees (EC), to make sure that all participants in the trials are safe. The EC includes scientists, healthcare providers, and people in the community. The EC reviews, approves, and watches over all research for humans in that hospital; to make sure the rights of trial volunteers are protected. This is the law.
The EC review makes the trials as safe as possible but there are always still some risks. When the trials are for people who are very sick, the risks are usually higher. With all trials, information will be given about ways that the treatment might be helpful or harmful. This information is part of the “informed consent form” (ICF). The ICF will be reviewed with you by the trial doctor (Investigator), in your native language, to help you to decide to volunteer for the trial or not. The ICF will also tell you how to let the Investigator know if you think you are having a problem during the trial. You should ask any questions you may have about a clinical trial before signing the ICF. Even after you have signed the ICF to volunteer in a clinical trial, you can always speak to the Investigator or any trial team member, at any time during the trial, if you have more questions or problems.
Also, there is a Data Safety Monitoring Board (DSMB) which is another group of people made up of doctors and other scientists not involved in the trial. The job of the DSMB is to look at and watch over the trial safety results. In case they believe that you might be harmed, they can stop the trial any time if:
- The new trial treatment might be more harmful than helpful.
- The results show that the new treatment is much better (or much worse), so that all trial volunteers get the better treatment.
NISCI stands for Nogo Inhibition in Spinal Cord Injury. After a SCI, the body makes a chemical that helps to clean up the injury. But sometimes, too much clean-up chemical is made by the body and that can slow down healing of the injury. Scientists have seen this happen in animals with SCI and have also discovered the chemical that can restart the healing of the SCI. The chemical produced by your body that slows down healing is called Nogo-A and the chemical that acts against it is called anti-Nogo-A antibody. In animal studies and an earlier human trial, injecting the anti-Nogo-A antibody into the new injury showed that injured nerves in the spinal cord started to possibly regrow. The earlier human trial has shown this treatment to be safe. The goal of the current trial is to compare the effect, of the treatment (anti-Nogo-A antibody) to standard treatment, on arms and hand movement of people with a SCI in the neck.
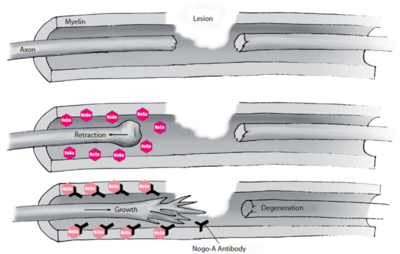
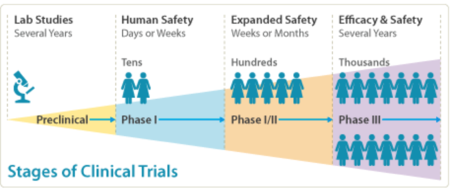
Scientists have been able to make a chemical like the chemical your body makes that acts against Nogo-A – this is called a monoclonal antibody and it is named anti-Nogo-A. Giving anti-Nogo-A after a SCI improved recovery in animals (preclinical studies). In a previous clinical trial, patients suffering from ALS (amyotrophic lateral sclerosis) who received a similar monoclonal antibody (NG-101) did not have problems after the treatment. This treatment is also now being used in patients with a form of MS (multiple sclerosis) and in animals that have had a stroke. In a previous clinical trial (NCT00406016; Phase I/IIa), NG-101 was given to 52 patients with complete SCI (from Germany, Switzerland and Canada) within 4-14 days of injury. No problems were reported in these trial patients after the treatment and so we are now going ahead with a larger clinical trial (the NISCI trial).
You will learn about this new treatment and are playing an active role in your own health care. You can help people who get a SCI in the future by contributing to SCI research now. To read about the risks of participation, please see 'Are clinical trials safe?'
At the beginning of the NISCI trial, a team of doctors, nurses and other healthcare providers at a trial centre will check your health and medical information to make sure that the trial is right for you. After signing the informed consent form (ICF) you will be included in the trial. The trial team will give you information about the treatment and medication which you will receive throughout the trial. The trial team will watch over you during the trial and will also stay in touch for some time even after the trial is completed. The NISCI trial follows a booklet of rules which includes information on the length of the trial and what information will be gathered.
Any time that you have a question, you can contact us through an email address or phone number that can be found on the NISCI website (www.nisci-2020.eu) or on our patient flyer. Our trial nurse/trial coordinators and the doctor will answer each question for you and/or family.
Yes. You can leave the NISCI trial at any time. Your participation in the NISCI trial is voluntary. You may decide not to participate at any time for any reason and without changes to your regular medical care. Also, your participation in the trial may be stopped at any time by the trial doctor or the Ethics Committee (EC) or the Data Safety Monitor Board (DSMB). Stopping the trial might happen if the risk is too high, if you are unable to follow the trial rules to make sure your are safe, and for any other reason that the trial doctor or committees thinks will make sure that you are not harmed.
If your participation in the trial is stopped by the doctor, the EC or the DSMB, you will still be asked to take the tests that usually take place at the end of the trial. If you stop participating for any reason or do not attend the final tests, we would still try to contact you to find out why.
Most clinical trials that compare a new treatment to a standard treatment will only be able to share results once the trial has been completed. If the trial is a "double-blind" trial as NISCI is, the doctor will not even know which treatment you received. The clinical trial rules will have the information about when the results will be available.
If this is something that you want to know more about, ask the trial doctor what the rules are before you agree to participate.
There will be no cost to you to participate. When a patient volunteers for and is accepted into the NISCI trial, the trial site provides the new treatment and any special tests and extra doctor visits that are required for the trial, at no cost to you.
Patients not living in one of the participating countries can participate in the trial if costs of hospitalization can be covered through insurance or other sources.
please visit the following website:






